 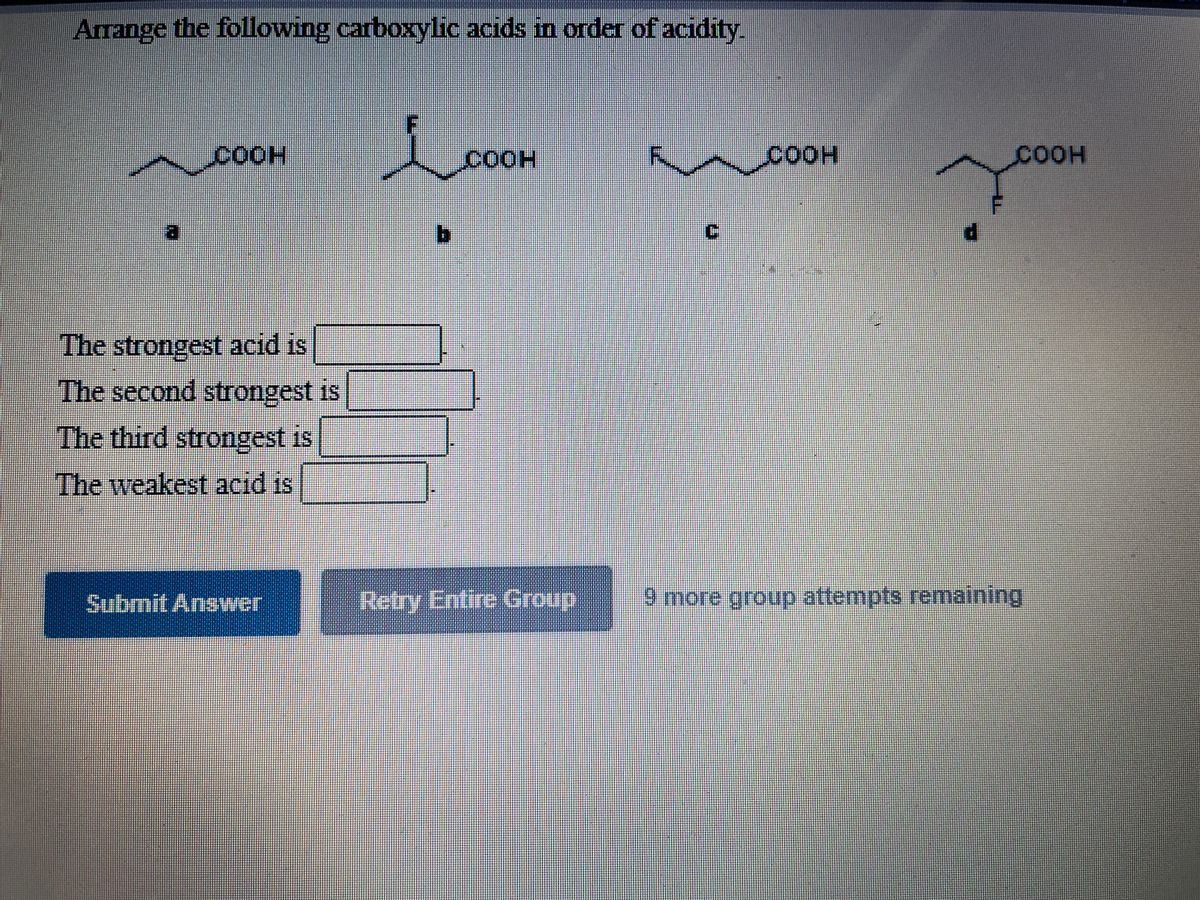 
They will be having Maximum authority due to the presence of two grooming groups and they will be showing negative. Oh the substitution that are linked to each compound, we can write the increasing order increasing order paul activity for the above compound as we will be having compound B. Asymmetric anhydrides (i.e., ones that decompose into two distinct carboxylic acids if hydrolyzed) can certainly be prepared. We have seen double bond here and always this is labeled as compounding now according to the according to the inductive effect. Can two carboxylic acids react The standard term for such molecules is acid anhydride, as they can be viewed as the product of a condensation reaction between two carboxylic acids, with concomitant loss of H2O. There are two bromine atoms linked together. And moving on to the structure of the last some pounds On the first carbon. Science Chemistry Chemistry questions and answers Arrange the following carboxylic acids in order of acidity. You have to keep in mind the order given in options and. Answer to Arrange the following carboxylic acids in order of. This problem is asking us to analyze these compounds and order them in order of decreasing acidity. The stability of the conjugate bases of respective acids should be compared and not the stability of the acids itself. Note: No matter the strength of the inductive effect, resonance is always stronger. Transcribed image text: Arrange the following carboxylic acids in order of acidity: Indicate, by letter, the strongest acid and the weakest acid of those. Double Bondo which and onto the veg bond here we have ch three group. So, the correct answer is Option D acetic acid < benzoic acid < formic acid. Moving on to the structure of the second compound that we have here we have double bond poet basically all the compounds are carb oxalic acid groups, compounds they contain the groups of delicacy. (b) Arrange the following in the increasing order of their reactivity towards nucleophilic addition reaction: C 6 H 5 COCH 3, CH 3 CHO, CH 3 COCH 3 (c) Why carboxylic acid does not give reactions of carbonyl group (d) Write the product in the following reaction (e) A and B are two functional isomers of compound C 3 H 6 O. It acts as an electron donating group, and so decreases the acidity of the carb. Arrange the following carboxylic acids in order of acidity: Find sample essays on related topics: Physics. So, um, are least acidic is going to be, uh, this third molecule with the metal group. Ask for homework help with other questions and get the answer fast StudentShare. The pK a s of some typical carboxylic acids are listed in the following table. This is the structure of the first compound here we have a witch labor last compound and on the second carbon we have Sinai group linked towards the dash bond. Answers for 1)Arrange the following carboxylic acids in order of acidity. In this question we are given with views of compound for which we have to write the correct increasing order of authority of the compound. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
